[FIN]CROSS-BORDERVOL: $4.2T
[SEC]CYBER ALERT: TIER2
[POL]IS0 GROWTH:+14%
[GEO] CLOUDINDEX: +2.4%
Structural Logic
Category Filters
Pharmaceutical Packaging Materials: How to Reduce Compliance Failure Risks
Lead Author
Published
Views:
For quality control and safety teams, pharmaceutical packaging materials are more than a supply item—they are a frontline compliance risk. From material compatibility and contamination control to traceability and regulatory documentation, one weak link can trigger costly failures. This article explains how to evaluate packaging systems more effectively and reduce compliance failure risks through smarter testing, supplier oversight, and standards-based decision-making.
Why do pharmaceutical packaging materials fail compliance reviews so often?
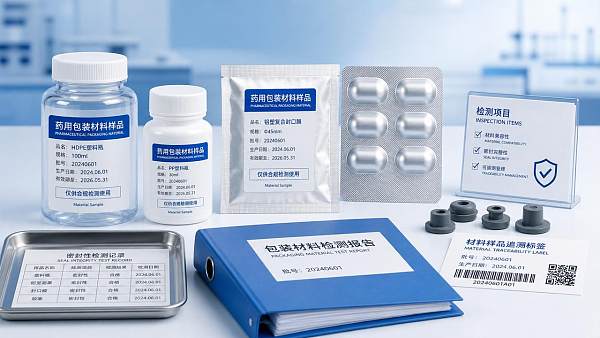
Compliance failures rarely come from a single dramatic defect. In most cases, pharmaceutical packaging materials fail because routine controls were fragmented across procurement, quality assurance, production, logistics, and document management.
For quality control personnel, the real challenge is not just identifying a nonconforming bottle, blister, film, stopper, or carton. It is proving that the full packaging system remained suitable, traceable, and stable throughout product life.
In a cross-industry environment, this problem is becoming more complex. Packaging risk now intersects with digital traceability, outsourced testing, supplier globalization, and faster product launches. Safety managers must evaluate not only materials, but also the data discipline surrounding them.
- Material changes may be introduced by suppliers without sufficient change notification or revalidation support.
- Incoming inspection may focus on appearance and dimensions while missing extractables, barrier shifts, or seal integrity risks.
- Batch records and certificates may exist, but they may not be linked in a way that supports rapid audit response.
- Packaging selected for cost efficiency may perform poorly under transport stress, sterilization, or long shelf-life conditions.
This is where a standards-based intelligence approach matters. G-MST supports decision-makers by connecting packaging evaluation with broader technical governance, supplier qualification logic, and testing, inspection, and certification workflows that reduce blind spots before an audit or deviation occurs.
What should quality teams check first in pharmaceutical packaging materials?
When reviewing pharmaceutical packaging materials, quality teams should begin with a risk-ranked framework rather than a generic checklist. Primary packaging, secondary packaging, printed components, and closure systems do not carry the same compliance consequences.
Core evaluation dimensions
- Product compatibility: Confirm that the material will not react with the formulation, absorb active ingredients, or release harmful substances over time.
- Barrier performance: Evaluate moisture, oxygen, light, and microbial protection according to product sensitivity and shelf-life requirements.
- Functional integrity: Review seal strength, torque retention, puncture resistance, breakage risk, and child-resistant or tamper-evident features where relevant.
- Cleanliness and contamination control: Assess particulate control, bioburden expectations, and manufacturing environment controls for packaging components.
- Traceability and documentation: Verify lot coding, certificates of analysis, change control notifications, and linkage to internal ERP or QMS records.
The following table helps quality and safety teams prioritize review points based on compliance impact rather than treating all packaging attributes equally.
A practical lesson from this matrix is clear: many compliance failures begin as documentation or change-control gaps, but they escalate because the material itself was not technically challenged early enough.
How to compare packaging options without creating hidden regulatory risk
Procurement teams often compare pharmaceutical packaging materials on unit price, lead time, and availability. Quality teams need a broader comparison model. A cheaper material may increase testing burden, deviation risk, and regulatory exposure.
The most effective comparison process combines technical performance, documentation maturity, supplier responsiveness, and lifecycle risk. This is especially important when sourcing across regions or using contract manufacturers with different quality cultures.
The table below compares common packaging decision factors that directly affect compliance failure risk in pharmaceutical packaging materials.
A lower quoted price is not a lower total risk. For safety managers, the stronger choice is usually the one that shortens investigations, withstands audits, and keeps release timelines stable under change.
Which standards and compliance records matter most?
Not every market applies the same documentation expectations, but quality teams should maintain a common core. Pharmaceutical packaging materials should be supported by a structured evidence package that aligns with product risk, region, and intended use.
Typical documentation baseline
- Material specifications with controlled revision history and approved test methods.
- Certificates of analysis or conformance linked to delivered lots.
- Declarations regarding relevant pharmacopeial, food-contact, or chemical substance restrictions where applicable.
- Extractables or chemical characterization support for high-risk primary contact materials.
- Supplier quality agreements, change notification rules, and complaint investigation procedures.
For companies operating across multiple sectors, G-MST’s advantage is the ability to connect packaging evidence with wider compliance infrastructure. That includes supplier master data, digital document workflows, and third-party testing visibility through TIC-oriented governance.
Why cross-functional traceability matters
A strong packaging file is no longer just a folder of PDFs. Quality and safety teams increasingly need searchable, linked evidence that connects approved specifications, incoming inspection results, deviation records, and released production batches.
This is particularly important during recalls, complaint trends, or regulatory inspection. If your team cannot rapidly prove what material was used, who approved a change, and what tests support that decision, the compliance risk rises sharply.
How to build a lower-risk supplier qualification process
A common mistake is treating pharmaceutical packaging materials suppliers like general commodity vendors. In reality, supplier qualification should be risk-based, evidence-driven, and tied to the criticality of the packaging component.
A practical qualification workflow
- Classify the component by product contact level, route of administration, and stability sensitivity.
- Review technical files, process controls, and available compliance declarations before commercial onboarding.
- Conduct sample evaluation under real filling, sealing, storage, and transport conditions.
- Assess supplier change-control discipline, complaint handling speed, and batch traceability practices.
- Define periodic review triggers such as formulation updates, repeated deviations, plant transfers, or raw material substitutions.
Quality teams often ask whether on-site audits are always necessary. The answer depends on component criticality and supplier maturity. For high-risk primary packaging, a stronger audit or third-party verification model is generally justified.
G-MST’s institutional value is especially relevant here. By integrating technical intelligence, procurement context, and TIC service awareness, it helps organizations avoid siloed qualification decisions that look acceptable on paper but fail under operational pressure.
What testing strategy reduces compliance failure risk fastest?
The fastest way to reduce risk is not necessarily adding every possible test. It is selecting the right tests at the right stage for the right failure mode. Pharmaceutical packaging materials should be tested according to component function and product vulnerability.
The table below maps practical testing priorities that quality control and safety teams can use during qualification and ongoing monitoring.
A targeted testing strategy saves time because it matches effort to failure probability. It also improves internal credibility, since quality teams can explain why a test was selected instead of appearing to overtest without a risk rationale.
Where do quality teams make the biggest mistakes?
Most avoidable failures come from assumptions. Teams assume a known supplier has not changed resin grade. They assume a secondary package cannot affect compliance. They assume prior approval means continued suitability.
Common misconceptions about pharmaceutical packaging materials
- “If the dimensions match, the component is acceptable.” Dimensional conformity does not confirm chemical or barrier equivalence.
- “Secondary packaging carries low risk.” Incorrect labels, poor print durability, or carton failure can trigger major traceability issues.
- “Supplier certificates are enough.” Certificates are useful, but they do not replace internal risk review and targeted verification.
- “A lower-cost alternate is easy to approve.” Even minor packaging substitutions may require stability assessment, line trials, and document updates.
For safety managers, the lesson is simple: treat packaging changes with the same discipline applied to other controlled product inputs. The cost of late discovery is usually higher than the cost of early scrutiny.
FAQ: practical decisions for pharmaceutical packaging materials
How should we prioritize testing when budgets are limited?
Start with the highest patient and product risk: primary contact materials, closure integrity, barrier performance, and traceability-critical printed items. Budget control should come from risk ranking, not from skipping evidence blindly.
Are pharmaceutical packaging materials from two suppliers interchangeable?
Not automatically. Even when dimensions appear equivalent, resin composition, additive packages, seal behavior, and change-control maturity may differ. Dual sourcing works best when specifications and qualification evidence are harmonized from the start.
What should procurement ask before approving a new packaging supplier?
Ask for controlled specifications, lot-level documentation, change notification rules, sample support, test capability details, and expected lead times for deviations or investigations. Procurement should also ask how quickly the supplier can support regulatory or audit questions.
How long does qualification typically take?
Timing depends on component criticality, required testing, and whether line trials or stability support are needed. Simple secondary items may move faster, while primary packaging materials often require a more structured review cycle before approval.
Why choose us when compliance risk is tied to packaging decisions?
G-MST brings a practical advantage for quality control and safety teams working with pharmaceutical packaging materials: it connects technical evaluation, supplier intelligence, and compliance-oriented decision support in one framework.
Instead of treating packaging as an isolated purchasing topic, we help organizations assess it as part of a larger operational system involving TIC services, digital records, procurement timing, and global regulatory movement.
You can consult us for
- Parameter confirmation for barrier, compatibility, seal, or documentation review priorities.
- Packaging material selection support when comparing multiple suppliers or substitute options.
- Delivery timeline planning for samples, qualification stages, and cross-functional approval workflows.
- Customized evaluation frameworks that align procurement, quality, safety, and audit-readiness goals.
- Certification and compliance evidence mapping for markets with different technical expectations.
- Quotation discussions tied to testing scope, supplier screening depth, and project urgency.
If your team is reviewing new pharmaceutical packaging materials, managing a supplier change, or trying to reduce compliance failure risk before the next audit, a focused technical consultation can clarify what to test, what to document, and what to challenge before approval.
Tags
Recommended for You








































